Nyheder

Guldvurdering fra EcoVadis
EcoVadis har tildelt Greiner Group den internationalt anerkendte bæredygtighedsvurdering, og med guldvurderingen er Greiner nu blandt de øverste 5% i branchen på verdensplan i forhold til bære-dygtighed.
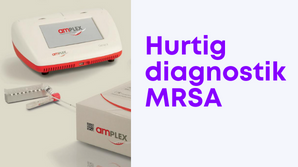
MRSA-test på 25 minutter
Med eazyplex® MRSA & MRSAplus kan du foretage in vitro-diagnostik, til påvisning af MRSA på 25 minutter med et bærebart apparat og testkits.
Læs mere
MycoXpert Mycoplasma Removal reagens (50x)
MycoXpert reagens er et antibiotikum, der kan tilsættes cellekulturmateriale.
Det er effektivt mod kontami-nering med Mycoplasma-arter.
Læs mere

Accuris Smart-Reader™ Multimode
Her er en avanceret mikroplade-læser designet til laboratorier inden for life science. Udstyret med højtydende optiske systemer er det en alt-i-en løsning til absorbans-, fluorescens- og luminescens-applikationer.
Læs mere
Her kan du følge med i vores nyheder, produktomtaler og guides. Vidste du for eksempel at vi blev ISO-certificeret i miljø- og kvalitetsledelse i 2022? Vi vil også gerne fortælle om nye brands, som for eksempel tyske Capricorn Scientific. Vi har nu et af de største udvalg af cellemedier og vi er særligt glade for, at det er europæisk producenter.
Nyhed: Biokeramiske 3D Scaffolds
Med Ossiforms P3D Scaffolds kan du skabe mere relevante vævs- og sygdomsmodeller, der fanger det komplekse samspil mellem forskellige celler.
Nyhed: Medie med reduceret serum
Har I en målsætning om at reducere forbruget af FBS? Med FBS Xtra får I en FBS med betydeligt reduceret mængde serum sammenlignet med traditionelt FBS.
Guide: Serumfri medier i cellekulturpraksis
Serumfrit medium kan være nøglen til mere avancerede cellekulturer og tilbyder samtidig en mere etisk løsning. I denne guide får du et overblik og link til serumfri medier.
Guide: Kom i gang med 3D-celledyrkning
Hvis du arbejder med celledyrkning og er interesseret i at udforske 3D-celledyrkning, vil denne guide giver dig et indblik i metoden og hvordan vi kan hjælpe dig.
Nyhed: Det nye In Vitro
Alle produktkategorier finder du nu i topmenuen under Produkter. Vi har ændret lidt i kategorierne for at gøre det lettere for dig at finde det, du leder efter.
Det er også i topmenuen, at du finder Produktguides, hvor du kan få overblik og blive inspireret. Ligeledes kan du blive klogere på vores primære leverandører under Brands og det er også i topmenuen, du kan tilgå Nyheder, hvor vi fortæller om nye produkter, brands og meget mere.
Nyhed: Capricorn Scientific
Vi er nu officielle distributører af Capricorn Scientifics produkter. Capricorn Scientific er en førende producent af cellekulturmedier, sera og reagenser af høj kvalitet til laboratoriebrug.
Med det nye samarbejde udvider vi vores produktportefølje og kan bl.a. tilbyde et bredere udvalg af serumfri, delvist serumfri og kemisk definerede medier.
Nyhed: 3x ISO-standarder
Siden 2020 har vi været i gang med en større udviklingsproces. Vision og strategi er blevet revitaliseret og vi arbejder målrettet på at blive en bæredygtig virksomhed.
I maj 2022 blev vi certificeret efter ISO-standarden 9001:2015, 13485:2016 og 14001:2015.
De tre certificeringer lægger en ramme for de krav og mål, vi skal overholde og dem vi har stillet os selv. Nu er vi på vej mod nye mål, godt hjulpet af de tre ISO-certificeringer.
Ja tak, jeg vil gerne modtage markedsføring på e-mail fra In Vitro A/S om Laboratorieudstyr og forbrugsvarer.
Det er til enhver tid muligt at frabede sig fremtidige henvendelser enten ved at kontakte In Vitro A/S eller ved at benytte frameldingslinket i udsendelsen.